Update (0859ET):
Hims & Hers Health fired back at Novo Nordisk's lawsuit on X, calling it a "blatant attack by a Danish company on millions of Americans" who depend on compounded medications to access personalized care.
HIMS continued:
Once again, Big Pharma is weaponizing the US judicial system to limit consumer choice. This lawsuit attacks more than just one medication or company – it directly assaults a well-established, vital component of US pharmacy practice that has improved patient care for everything from obesity to infertility to cancer. Hims & Hers has a long history of providing safe access to personalized healthcare to millions of Americans, and we will continue to fight to provide choice, affordability, and access.
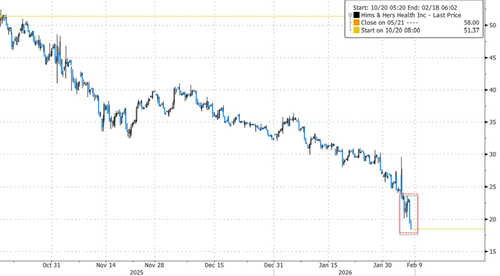
Shares of HIMS crashed in New York premarket trading, down about 20%, after the telehealth firm pulled its Wegovy copycat GLP-1 pill on Saturday. Novo hit the HIMS with a lawsuit earlier this morning.
* * *
The GLP-1 feud between Hims & Hers Health and Novo Nordisk keeps accelerating by the day.
The latest is that Novo is now taking legal action against HIMS, alleging that the telehealth firm "unlawfully mass markets unapproved versions of Novo Nordisk's FDA-approved semaglutide medicines, deceiving patients and putting their health at risk."
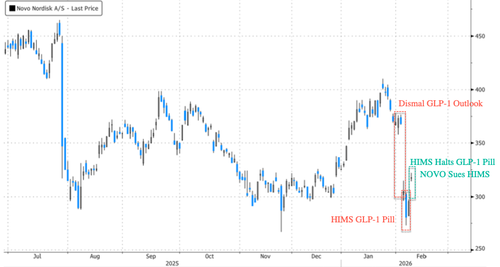
In late January, Novo released a $149 per month Wegovy pill, with HIMS just days later releasing a GLP-1 copycat pill of Wegovy for $49 a month.
By late last week, FDA Commissioner Marty Makary warned against companies "mass-marketing illegal copycat drugs, claiming they are similar to FDA-approved products."
Then, on Saturday, not even a day after Makary's comments, HIMS pulled the GLP-1 copycat pill ...
Back to Novo's lawsuit.
Here are the three takeaways:
-
Hims & Hers unlawfully mass markets unapproved versions of Novo Nordisk's FDA-approved semaglutide medicines, deceiving patients and putting their health at risk
-
Novo Nordisk takes decisive legal action to stop Hims' illegal conduct, protect public health, and defend the scientific innovations that deliver better health outcomes to Americans living with serious chronic diseases like obesity and diabetes
-
Novo Nordisk is asking the court to permanently ban Hims from selling unapproved, compounded drugs that infringe our patents, and is seeking to recover damages
"Hims & Hers is mass marketing unapproved knock-off versions of Wegovy and Ozempic that evade the FDA's gold standard review process – that's dangerous and deceptive to patients, and undermines the scientific innovation and regulatory rigor in place to ensure these treatments are safe and effective," John F. Kuckelman, senior vice president, Group General Counsel, Global Legal, IP and Security, wrote in a statement.
Kuckelman noted, "We've taken legal action to protect the American public and our intellectual property and will continue to work with regulators, law enforcement, and other key stakeholders to ensure patients have access to FDA-approved safe and effective medicines."
In markets, HIMS shares fell 20% in New York premarket trading after its GLP-1 copycat pill was pulled, with additional pressure from Novo's lawsuit.
On the other hand, Novo Shares in Denmark moved up 7%. Shares were beaten down last week amid a dismal full-year earnings outlook and HIMS' GLP-1 copycat pill.
Here's our coverage on the HIMS-Novo feud:
Citi analyst Daniel Grosslight told clients that the FDA's crackdown on HIMS' "aggressive compounding practices" suggests there could be further downside if the FDA "completely eliminates or severely curtails" HIMS' ability to compound GLP-1s. He rates the stock "Sell" and slashed his price target to $16.50 from $30.